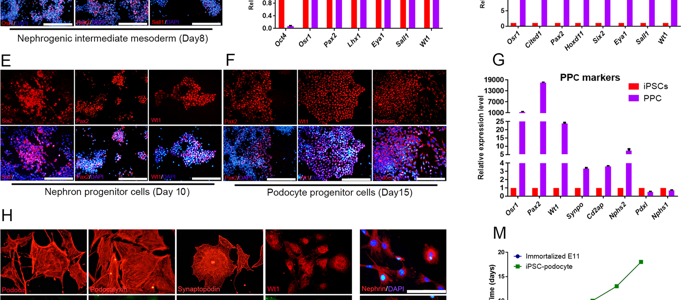
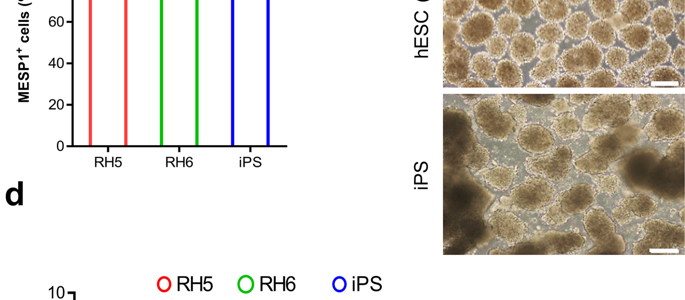
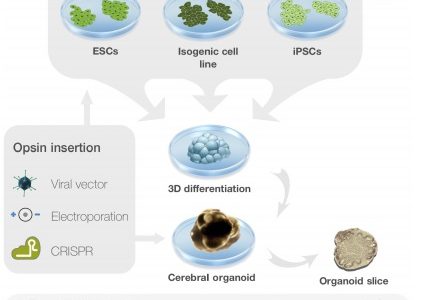
It is a great pleasure to invite you all to attend the 16th Royan Virtual International Congress on Stem Cell Biology and Technology which will be held on September 5-6, 2020. The congress is part of the 5-day virtual Royan International Twin Congress and is accompanied by the 21st Congress on Reproductive Biomedicine.
This event shares an insight into
the recent and cutting edge advances in the fields of stem cells and
regenerative medicine. The goal is to provide a unique platform for
researchers, engineers, clinicians and students to present and exchange
break-through ideas to promote interdisciplinary research.
Due to the COVID19 pandemic
across the world, the congress has been converted to a fully virtual event. We
have invited renowned scientists from different countries around the world.
Please visit us at www.royancongress.com to find
more information on the speakers, abstract submission and etc.
We look forward to welcome you to this exciting event.